|
When the tarsition of the Electron takes place any given higher Orbit with the principal quantum number starting from n= 5,6,7… to the fourth Orbit, then we obtain the brackett series of the Hydrogen Atom. These are obtained at the infrared region. to the third Orbit, then we obtain the paschen series of the Hydrogen Atom. When the transition of the Electron takes place from any higher Orbit with the principal quantum number starting from n=4,5. These are obtained at the visible region. When the transition of the Electron from any higher Orbit, the principal quantum number starting from n=3,4,5… to the second Orbit with the principal quantum number n=2, we obtain the balmer series of the Hydrogen Atom. This is said to be obtained in the ultraviolet region. When the transition of the Electron takes place from any given higher Orbit to the first Orbit, we obtain the lyman series of the Hydrogen series. These series obtained are labelled as Lyman series, Balmer series, Paschen series, Brackett series, Pfund series, Henry series. This indicates that the different transitions witnessed give rise to different series of lines. It is very important to note that each transition corresponds to a particular and definite characteristic wavelength. The vertical lines on the other hand refer to the transition that the Electron undergoes moving from the higher Energy level to the lower Energy level. The horizontal lines witnessed refer to the different Energy levels. The Energy level diagram for the Hydrogen Atom indicates the different series of lines that is observed in the Spectrum. They were an Atomic fingerprint that resulted from the Atom’s internal structure. Several experiments have proved that the wavelengths of the lines were given as the characteristic of the chemical element that emits the light. In the late 1800s, it was referred to as when gas is excited with an Electric discharge and the light emitted can be viewed using a diffraction grating the observed Spectrum contains not of a continuous light band, however, of individual lines with the wavelengths, that are well-defined. 
This Radiation emitted is in the form of the Spectral lines. The v in hv denotes the frequency of the Radiation Energy. In order to gain back the stability, the Electrons jump from the higher Orbits to the lower Orbits which results with the emission of the Energy possessing the value hv. When the Electrons start revolving around these excited states, then the Atom is claimed to become unstable. The given Electron gains Energy from its surroundings that results in its jumping into higher Orbits possessing the principal quantum number n= 2, 4, 4, 5… these higher Orbits are called the excited states. The Hydrogen Atom is claimed to be stable when the Electron that is present in it starts revolving around the nucleus present in the first Orbit with the quantum number n=1. The origin of the Spectral lines associated with Hydrogen can be explained with the help of Bohr’s theory. This overall phenomenon accounts for the emission Spectrum via Hydrogen, too, better referred to as the Hydrogen Emission Spectrum. They also emit Radiation when they return to their original states. 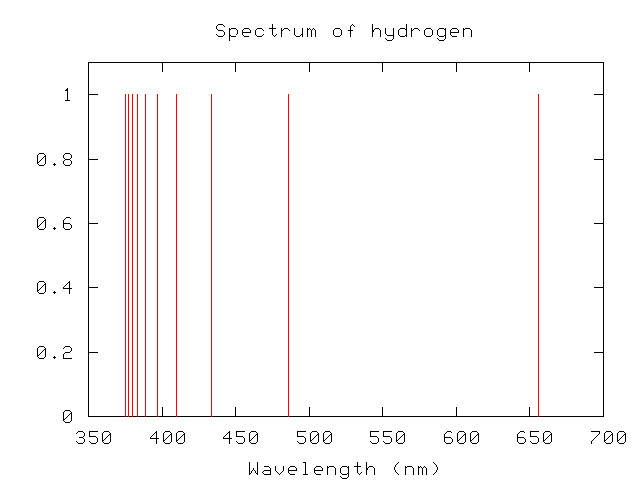
They will jump from a lower Energy level to a higher Energy level. 
We all know that the Electrons present in either a Molecule or an Atom absorb the Energy and get excited.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
